ABSTRACT
Introduction: Self-diagnose and obtaining various health products via the Internet were extremely risky practices as it may increase the risks of patient injury or even death. Objective: This study aims to explore the patterns of prescription medicines sale through e-marketplace (specifically: Shopee) in Malaysia as well as the associated factors. Method: Cross-sectional study secondary data of 983 Advertisement Screening Reports (reported from 1st January 2020 – 30th June 2020) in Johor State were collected. Descriptive statistics using frequency, percentages and/ bar charts using Microsoft Excel 2013 were used to report on the patterns of prescription medicines sale through e-marketplace (specifically: Shopee) in Malaysia based on types of prescription medicines that are being advertised, registration status of prescription medicine that are being advertised as well as frequently violated Malaysia’s law related to prescription medicine by sellers on Shopee. Factors associated were explored by Simple and Multiple Logistic Regressions using IBM SPSS Version 22. Results and Discussion: 796 out of 852 (93.4%) prescription medicines samples were not registered with the Drug Control Authority, Malaysia’s Health Ministry. Hormones (62.6%, while sex hormones which were anabolic steroids showed the highest frequency; 58.3%) were the highest prescription medicines sold through the e-marketplace in Malaysia, followed by antibiotics, clomiphene (fertility drug), sibutramine (slimming pill) and prostaglandin and its synthetic derivatives (abortion pill) denote 4.8%, 2.2%, 1.8%, 0.4% respectively. The unregistered prescription medicines were found to be the highest to violate Section 13(a) of Poison Act 1952 which include 766 samples. Multiple logistic regression tests indicate that sellers who violated Regulation 7(1)(a) of Control of Drug and Cosmetic Regulations 1984 (Adjusted OR: 728.63; 95%CI; 194.69, 2726.96%; p = 0.000) have higher chance to sell unregistered medicines through e-marketplace compared to those who violated Section 13 (a) of Poison Act 1952 (Adjusted OR: 0.012; 95%CI; 0.002, 0.058%; p = 0.000), and Section 4B of Malaysia Advertisement and Sale Act 1956 (Adjusted OR: 0.060; 95%CI; 0.014, 0.260%; p = 0.000). Conclusion: The findings in this study may give a brief idea for improving the current practice in order to curb the freely illegal prescription medicines sale through e-marketplace (specifically: Shopee) without the supervision of professionals.
INTRODUCTION
In the era of the internet, the ways of getting treatment for any disorder or disease have been revolutionized among the public. People now use the internet not only for retrieving health information, but also to self-diagnose and obtain various health services or products [1]. The expansion of the internet services has made the online sale of prescription medicines largely available to patients or consumers through e-commerce platforms. However, there is an increased risk of patients buying products (which dominate the global online pharmacy market) from illegal sites [1].
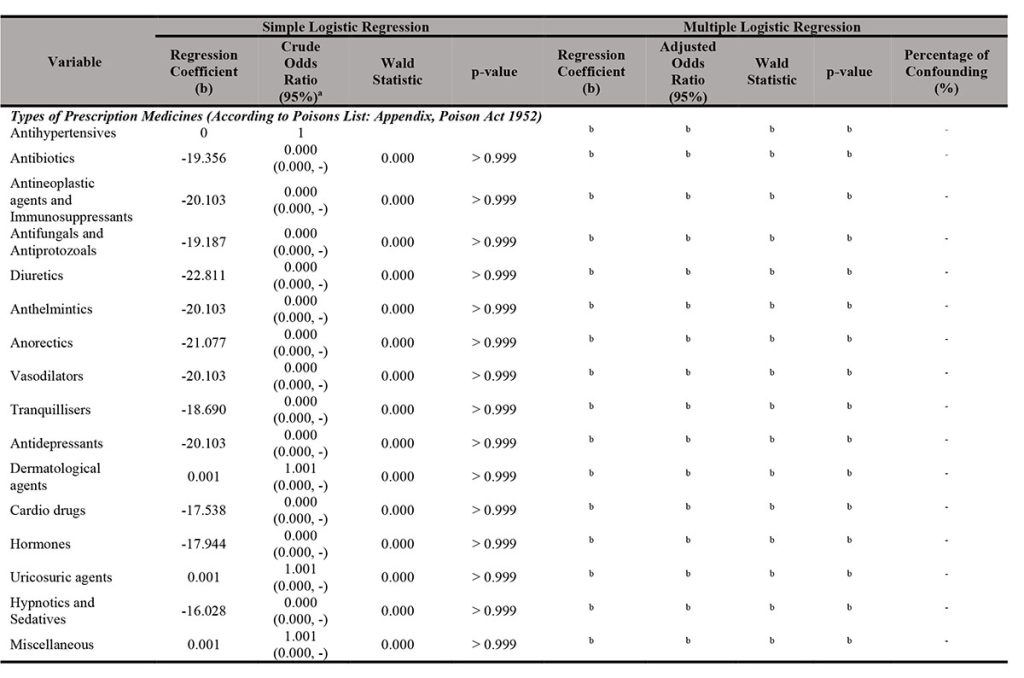
In Malaysia, prescription medicines are listed as Group B (Part 1) in the First Schedule of the Poisons Act 1952 which classifies them as poisons. This is because these prescription medicines have very specific treatment properties; therefore, the administration, sale, and supply of these substances as dispensed medicine are limited to a specific group of professionals namely registered medical practitioners, registered dentists (division I), and veterinary surgeons. Licensed pharmacists can sell or supply prescription medicines only on a prescription provided by the mentioned groups of people. Such requirements for the supply of prescription medicines are needed because these medicines can pose a danger if sold or supplied without proper diagnosis or dispensing. Additionally, all drugs (prescription-only and over-the-counter) and even dietary supplements can cause harm if taken without appropriate advice and supervision of a medical doctor or pharmacist [1]. Patients can be exposed to the dangers of toxicity, under-dosing, contraindications, and various side effects including failure of vital organs such as kidneys, lungs, and heart. The most important concerns are inadequate or inaccurate information about the patient’s health status and medications, inappropriate self-diagnosis, or incomplete management of drug-related issues, such as polypharmacy, therapeutic duplications, adverse events, and drug-to-drug or drug-herbal interactions [1].
While legitimate health practitioners and pharmacies provide many benefits including convenience and privacy, the public in general also experiences some benefits when purchasing prescription medicines via the internet. However, the proliferation of illicit online selling of prescription medicines are their associated risks of patient injury or even death and thus, the internet is an extremely dangerous practice [2]. A study in 2002 found that e-pharmacies are selling anabolic steroids, human growth hormone, drugs of addiction and antibiotics, which are supplied in inappropriate quantities and dosage without prescriptions and without medical consultations. E-pharmacies also provide unfitting promotion of medicines, poor packaging and handling practices. These may potentially lead to inappropriate medication use, adverse events and could worsen global problems such as antibiotic resistance [3].
In 2019, Pharmacy Enforcement Division of Malaysia’s Health Ministry issued notices to e-commerce platforms to demand the removal of links that advertise illegal pharmaceutical products [2]. Since 2016, nearly 6,000 of such notices were sent out, but most of them, numbering to 4579, were issued in 2018 [2]. However, the exact number of online pharmacies and consumers buying prescription medicines online, the volume of drugs traded, and the revenue and profits generated by such a hidden business are very difficult to estimate [4].
Malaysia’s Health Ministry also highlighted that some medicines which are copiously being sold on e-commerce platforms are found to be adulterated with banned substances or poisons and warned that they might be fake [5] or counterfeit health products which may contribute to patient’s safety risks [1]as well as support abuse of prescription drugs [4]. Indeed, the World Health Organization (WHO) found that 10% of medicines sold globally through online suppliers may be counterfeit [6], ranging from less than 1% in the developed countries to over 30% in developing countries such as Africa, Asia, India, and Latin America [1]. Furthermore, medications used for both acute and chronic conditions can be freely purchased by users of online pharmacies without ensuring whether they are legitimate sites [1]. Moreover, not only illegitimate online pharmacies pose a definite threat to patients through the marketing of counterfeit medications and substandard products, but legitimate sites may also pose similar issues [1].
A study in 2002 by Bessel found that the country of origin could not be identified for 22 websites which e-pharmacies had operated in at least 13 countries [3]. This study expressed that it was difficult to identify the country of origin from which the medicines were sold [3]. Bessel et al. also found that 30% of the 104 e-pharmacies delivered nationally and 67% internationally [3]. This showed that the geographical distribution of the phenomenon seemed to be very heterogeneous [4]. This study noted that safeguarding consumers and ensuring the quality of websites that sold medicines across state and national boundaries were both complex and difficult [3].
In a pilot study at a mail centre in Carson City, California, US customs officers found 721 parcels (from a total of 1908 suspicious parcels inspected) containing 197 different types of medicines which came from 19 different countries, including medicines of potential addiction and abuse, antibiotics, steroids and medicines with narrow therapeutic indices primarily obtained via internet sale [1]. It was estimated that approximately two million parcels containing scheduled medicines for personal use entered the United States of America (USA) each year, which placed an overwhelming burden on US customs [3].
More worrying, the study by Bessel et al. (2002) addressed that 19% of websites which denote 20 websites from a total of 104 e-pharmacies appeared to supply prescription-only medicines with no prescription and only 12% of e-pharmacies displayed quality accreditation seals [3]. Thirty seven percent of websites were found with no privacy statements, information disclaimers, and return policies, whereby e-pharmacies display that such policies often place all risk and responsibilities with the consumer and little with the site operator [3]. Although 42% of websites promoted the availability of advice from pharmacists, it is difficult to determine whether registered pharmacists were involved [3]. Twenty percent of 104 e-pharmacies promoted prescription-only medicines, 19% non-prescription medicines, 20% complementary medicines and 23% health and beauty products [3].
Consumer behaviour may be altered and prescription medicines use may be stimulated inappropriately and unnecessarily due to direct-to-consumer advertising of medicines [3]. The e-pharmacies appeared to undermine the safe and appropriate use of medicines because the good practice, organization, and support functions which are usually considered necessary to provide quality of care were largely absent [3].
In Malaysia, e-commerce platforms such as Lazada, Shopee, PrestoMall, Mudah.my, Qoo10.my, Lelong.com.my, Jamumall.com, and Youbeli.com are the few platforms which were found to be liberally selling prescription medicines. As of the first quarter of 2020, Shopee led as the most clicked e-commerce site in Malaysia, followed by Lazada and Lelong [2]. Shopee was the leading e-marketplace not only in Malaysia, but also across Southeast Asia with approximately 27 million clicks [2].
Currently, some of the jurisdictional issues in cyberspace are being addressed, especially pertaining to fraud, paedophilia, and gambling; and these outcomes may also apply to the delivery of e-health services [3]. The solutions to combat illicit online selling of prescription medicines lack focus on innovative technologies and the need for harmonised nationwide regulations that evolve rapidly with the evolution of e-commerce and internet technology [2]. However, most current drug policies, legislation, and practice standards which were devised by many countries with the purpose of supporting the safe and appropriate use of medicines were written without reference and prior to the evolution of electronic technologies and online sale of medicines [3].
Also, the national regulatory measures do not protect the global public from poor practices involving online sale of medicines as the internet operates independently of geographic boundaries, which results in difficulty to establish legal jurisdiction in a global online environment (which includes consumer protection mechanisms such as privacy statements, information disclaimers, and return policies) [3]. The study by Bessel et al. (2002) demonstrated that a wide range of medicines could be bought from many countries around the world, including countries that do not have adequate regulatory frameworks for the safe and appropriate use of medicines [3]. Moreover, one cannot be certain of the regulatory framework under which the internet pharmacy is operating since many illegitimate websites are unwilling to indicate their actual location [1]. Thus, the inadequate regulatory frameworks or resources to safeguard consumers in some countries may lead to consumers being put at risk when they buy medicines online [3]. Bessel et al. added that self-regulation may be ineffective due to the inherent global nature of the internet [3]. The national authorities are often powerless beyond their borders which may further complicate the issue [1].
There is a need to build nationwide consensus for a coordinated multi stakeholder response to address the unique and complex threats posed by unlawful online selling of prescription medicines in order to ensure the safety of patients globally [4]. Until today, there is no published study done locally or internationally regarding patterns of prescription medicine sale through the e-marketplace and the associated factors. Nonetheless, there is one [1] study conducted in 2015 in the United States which found that the numbers of prescription medicines online sale were highest for neuropsychiatry therapeutic area (20.27%) followed by cardiovascular disease therapeutic area (18.86%) and infectious disease therapeutic area (16%) [7]. However, this study did not specifically focus on the e-marketplace. Another study in 2018 found that most of the therapeutic categories of drugs were freely available and could be purchased online, which ranged from the performance and image-enhancing “lifestyle drugs” (such as phosphodiesterase type 5 inhibitors, anti-baldness products, obesity products) as well as life-saving medicines (such as analgesics which include nonsteroidal anti-inflammatory drugs and opioids, psychiatric, and cardiologic drugs) [1]. This illegal market facilitated the trading of apparently identical products in an uncontrolled situation, with no restrictions on the consumers (e.g., people under 18 can also purchase medications online) or on products (larger quantities can be purchased) from a large virtual supply [1].
Therefore, the aim of this study is to explore the patterns of prescription medicine sale through e-marketplaces that can be accessed from www.shopee.com.my and the Shopee application in Malaysia. The patterns that were observed include registration status of prescription medicine sale, types of prescription medicine sale, and frequently violated Malaysian laws related to prescription medicine by sellers on Shopee. Furthermore, the study is also intended to identify the associated factors with the patterns of prescription medicine sale through the e-marketplace (specifically: Shopee) in Malaysia (i.e., types of prescription medicine and Malaysian laws related to prescription medicine that are frequently violated). The study aims to serve as the gateway for decision makers on deciding the improvement in the legal system.
METHOD
Study Design
This is a cross-sectional study. Data were collected retrospectively from Advertisement Screening Reports written by Pharmacy Enforcement Officers in Johor State from 1st January 2020 – 30th June 2020. Beginning in 2020, the Pharmacy Enforcement Branch of Johor State had been assigned by the Pharmaceutical Service Division, Ministry of Health Malaysia to review the online selling of prescription medicines on Shopee. Thus, prescription medicines that were being sold on Shopee were reviewed based on online shops registered on Shopee. All the prescription medicines that were being sold from the online shop identified were thoroughly screened. Information on the prescription medicine screened was collected by filling up the ‘Borang Saringan Iklan’ which is available in ‘Garis Panduan Penyaringan Iklan’, the guideline set by the Pharmaceutical Service Division, Health Ministry, Malaysia. Generally, the Advertisement Screening Reports studied have a snapshot of the advertisement on Shopee which is attached with the ‘Borang Saringan Iklan’, product registration status search, and advertisement approval search. All the advertisement screening reports approved by the Head of Pharmacy Enforcement Branch of Johor State were studied retrospectively.
The patterns of prescription medicine sale through e-marketplace (specifically: Shopee) in Malaysia were gleaned by obtaining secondary data from the Advertisement Screening Reports on (i) Registration Status, (ii) Types of Prescription Medicines, and (iii) Malaysian Laws Related to Prescription Medicine that are Frequently Violated. The secondary data was screened and reviewed by different researchers to ensure accuracy. The secondary data included was from January till June 2020. Shopee was selected in this study because the Johor Pharmacy Enforcement Branch was instructed by the Pharmaceutical Service Division of Malaysia’s Health Ministry to review the online selling of prescription medicines on Shopee. Additionally, Shopee led the Malaysian e-marketplace as the most clicked site in the country as of the first quarter of 2020, and as the leading e-marketplace not only in Malaysia, but also across Southeast Asia [2].
The patterns of prescription medicine sale through e-marketplace (specifically: Shopee) were depicted in a 6-month study on the selling frequency of prescription medicines through e-marketplace in Malaysia based on (i) Registration Status, (ii) Types of Prescription Medicines, and (iii) Malaysian Laws Related to Prescription Medicine that are Frequently Violated. Kindly refer to Appendix 1 for the Data Collection Form.
The applicability of prescription medicine sale through e-marketplace (specifically: Shopee) requires further knowledge on the relationship (i.e., possible factors) of the prescription medicine sale through e-marketplace in Malaysia with (i) Types of Prescription Medicines, and (ii) Malaysian Laws Related to Prescription Medicine that are Frequently Violated. These factors were identified via further statistical analyses.
Study Population
All Advertisement Screening Reports which were reported by Pharmacy Enforcement Officers in Johor State from 1st January 2020 – 30th June 2020 were included in this study.
Inclusion Criteria
Advertisement Screening Reports with complete information on the (i) Registration Status, (ii) Types of Prescription Medicines, and (iii) Malaysian Laws Related to Prescription Medicine that are Frequently Violated which were reported by Pharmacy Enforcement Officers in Johor State from 1st January 2020 – 30th June 2020 were included in this study.
Exclusion Criteria
Advertisement Screening Reports with incomplete information on the (i) Registration Status, (ii) Types of Prescription Medicines, and (iii) Malaysian Laws Related to Prescription Medicine that are Frequently Violated which were reported by Pharmacy Enforcement Officers in Johor State from 1st January 2020 – 30th June 2020 were excluded in this study. Combination medicines which have more than one classification according to the Poison List Appendix of Malaysia’s Poison Act 1952 were excluded from sampling selection because they may complicate the analysis of the result. Group C poisons also were excluded because this study focuses on Group B poisons. The categorisation of Group B poisons and Group C poisons can be referred to the First Schedule, Poison List of Malaysia’s Poison Act 1952.
Study Duration and Timeline
The data was collected retrospectively from August till October 2020 from the Advertisement Screening Reports, which were reported by Pharmacy Enforcement Officers in Johor State from 1st January 2020 – 30th June 2020.
This timeframe was selected because of the high likelihood of online selling and shopping during this period of time due to the Covid-19 pandemic. Recent research in Bangalore (India) has shown that 50% of their consumers spent time searching for information and purchasing products via online platforms during the Covid-19 pandemic [4]. Other research in Malaysia showed that most sale and purchase activities were carried out online during the Covid-19 pandemic [8].
Study Procedures
The study procedures were simplified as in Figure I.
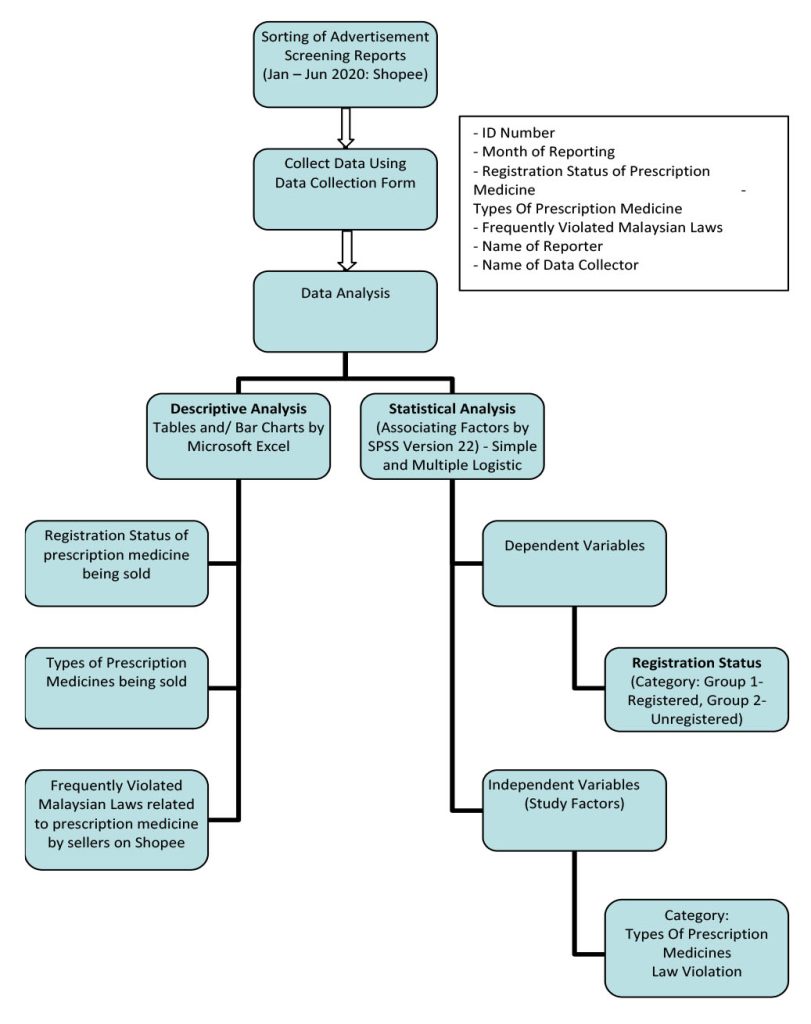
Grouping of Product Registration Status in Prescription Medicines Sale through Shopee.
The registration status of the medicine was identified through a search using the official website of the National Pharmaceutical Regulatory Agency (NPRA).
Grouping of Types of Prescription Medicines Sale through Shopee.
The grouping was done according to the Poison List Appendix of the Poison Act 1952, which categorises the medication types into several basic groups.
Grouping of Malaysian Laws Related to Prescription Medicines That Are Frequently Violated in Online Selling of Prescription Medicines through Shopee.
All the frequently violated relevant Malaysia’s laws related to prescription medicine were identified. Relevant Malaysia’s law related to prescription medicine that frequently violated concern here are Section 13(a) of Poison Act 1952 (Prescription medicines sold by unlicensed personnel), Regulation 7(1)(a) of Control of Drugs and Cosmetics Regulations 1984 (Prescription medicine that being sold is not registered with Drug Control Authority, Malaysia’s Health Ministry), Section 3(1) Medicine (Advertisement and Sale) Act (MASA) 1956 (Prohibition of advertisements relating to certain diseases listed in the MASA 1956), Section 4 MASA 1956 (Prohibition of advertisements relating to abortion), and Section 4B MASA 1956 (Advertisements of medicines without approval from Medicine Advertisements Board).
Statistical Analysis Plan
The study samples included 983 Advertisement Screening Reports that had been checked and approved by the Johor Enforcement Branch from January till June 2020. Statistical analyses consisted of two steps, namely descriptive statistics and multiple logistic regressions.
Descriptive statistics using frequency and percentages were used to report the patterns of prescription medicines sale through e-marketplace (specifically: Shopee) in Malaysia based on types of prescription medicines that were being advertised. Meanwhile, the patterns of prescription medicines sale through e-marketplace (specifically: Shopee) in Malaysia based on registration status of prescription medicines that were being advertised as well as frequently violated Malaysian laws related to prescription medicines by sellers on Shopee were presented in the form of bar charts using Microsoft Excel 2013. The difference in the bar charts provides an overview of prescription medicines sold through Shopee in Malaysia.
The data was further analysed using Simple (Enter Method) and Multiple Logistic Regressions models of SPSS Version 22 (Backward Elimination (LR) Method) to determine the factors associated with patterns of prescription medicines sold through the e-marketplace (specifically: Shopee) in Malaysia. The possible factors were determined based on the magnitude of relationship between registration status of prescription medicines sales with i) the types of prescription medicines that are being sold on Shopee and ii) frequently violated Malaysian laws related to prescription medicines that were measured by the adjusted odds ratio (OR) of the multivariable analysis. For inferring significance of the findings, the level of significance (α) was set at 0.05 and confidence interval of 95% with the product registration status as a reference. Besides, confounders also were identified by comparing the estimated measure of association before and after adjusting for confounding [9]. In other words, the measure of association was computed both before and after adjusting for a potential confounding factor [9]. If the difference between the two measures of association was 10% or more, then confounding was present [9]. If it was less than 10%, then there was little, if any, confounding [9].
The formula is shown below:
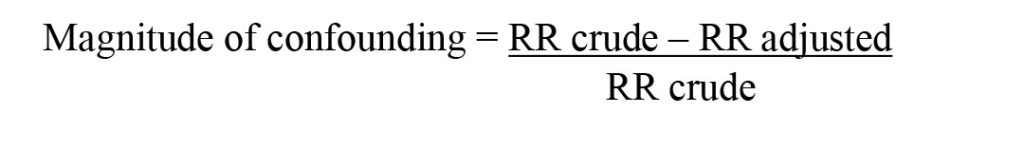
Where Relative Risk (RR) crude is presented as crude odds ratio (95%) obtained from a simple logistic regression test analysis, meanwhile RR adjusted is presented as adjusted odd ratio (95%) obtained from a multiple logistic regression test.
Study Outcome
The patterns shown by the bar charts and the result of statistical analyses of the relationship provided an overview on prescription medicines sale through e-marketplace (specifically: Shoppe) in Malaysia. The investigators hope that the results will initiate a discussion, specifically on how future pharmacy enforcement legislation should be implemented.
Ethics of Study
The Medical Research Ethics Committee (MREC), Ministry of Health Malaysia has determined that this study does not require MREC review/approval as this study does not involve any human subjects/intervention on human subjects. The MREC, and MOH allowed this study to be conducted without the need for further submission or reports to MREC.
Privacy and Confidentiality
Identification numbers (ID No.) instead of Sellers’ names were used on the Data Collection Form (Kindly refer to Appendix 1). All data were entered into a computer that’s password protected. On completion of the study, data in the computer were copied to compact discs (CDs) and data in the computer were erased. CDs and any hardcopy data were stored in a locked office of the investigators, to be maintained for a minimum of 3 years after the completion of the study. The CDs and data would be destroyed after this period of storage.
RESULTS
Descriptive Statistics: Patterns of Prescription Medicines Sale through E-Marketplace (Specifically: Shopee) in Malaysia.
Data extracted from the approved Advertisement Screening Reports resulted in 983 samples of prescription medicines sold through the e-marketplace (specifically: Shopee) in Malaysia. Then, 46 samples were excluded because they had more than one classification (based on the Poison List Appendix of Malaysia’s Poison Act 1952), which could have complicated the analysis of results, while another 85 samples were excluded because they were categorised as Group C (Part I) in the First Schedule of Malaysia’s Poison Act 1952. There was no data with missing information encountered during sampling. No incomplete reports were found in this study.
Registration Status of Prescription Medicines Sale
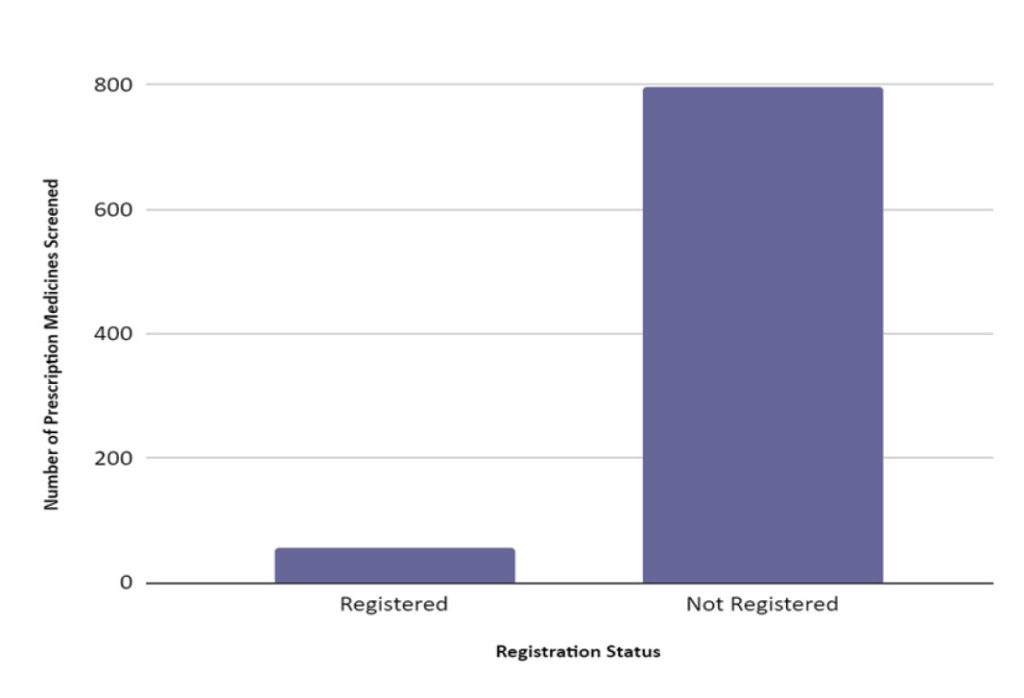
From a total of 852 approved samples, 796 (93.4%) prescription medicines were found not registered with the Drug Control Authority (DCA), Health Ministry, Malaysia and only 56 (6.6%) were found as registered (Figure II.)
Types of Prescription Medicines Sale
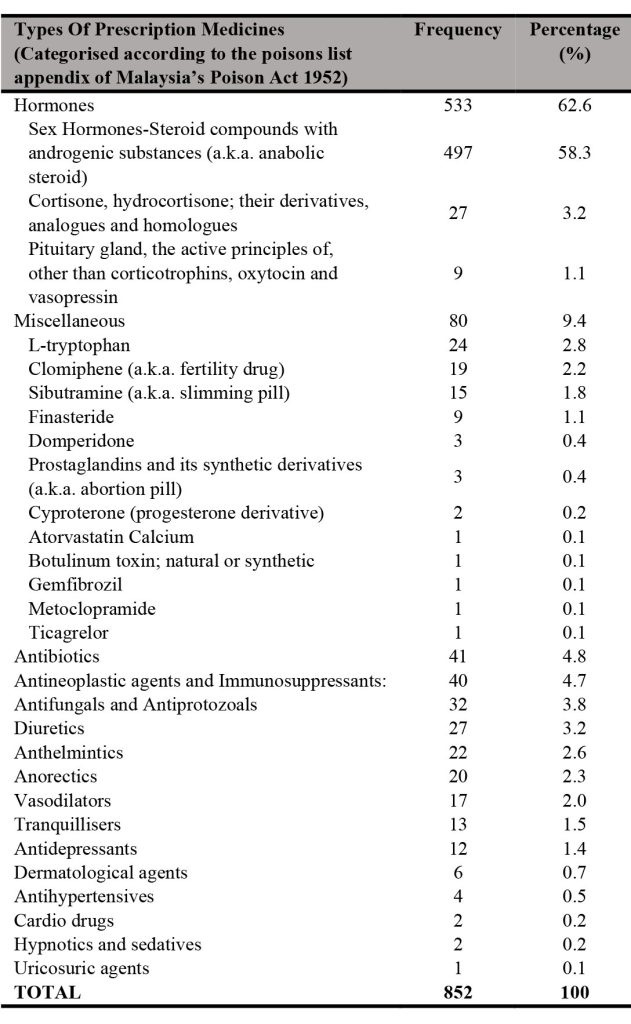
Table I shows that hormones have the highest sales with a total of 533, accounting for 62.6% of the total prescription medicines sold through e-marketplace in Malaysia.
The hormones were followed by the miscellaneous group (80 samples which account for 9.4%) and antibiotics (41 samples which account for 4.8% only). Antineoplastic agents and immunosuppressants also showed quite a high total (40 samples which account for 4.7%) for prescription medicines sold through e-marketplace (specifically: Shopee) in Malaysia. Sex hormones in the form of steroid compounds with androgenic components (i.e., commonly known as anabolic steroid) showed the highest frequency (497 samples which accounts for 58.3%) compared with other types of prescription medicines. Clomiphene which is a fertility drug and sibutramine which is a slimming pill were also found to be prescription medicines frequently sold with 19 samples (which account for 2.2%) and 15 samples (which accounts\ for 1.8%), respectively. More worrying, prostaglandins and their synthetic derivatives, which are found in abortion pills were also found to be sold with 3 samples (0.4%). These prescription medicines were found to be freely sold illegally through e-marketplace (specifically: Shopee) in Malaysia from January till June 2020 without supervision of professionals such as doctors and pharmacists.
Frequently Violated Malaysian Laws Related to Prescription Medicines by Sellers on Shopee
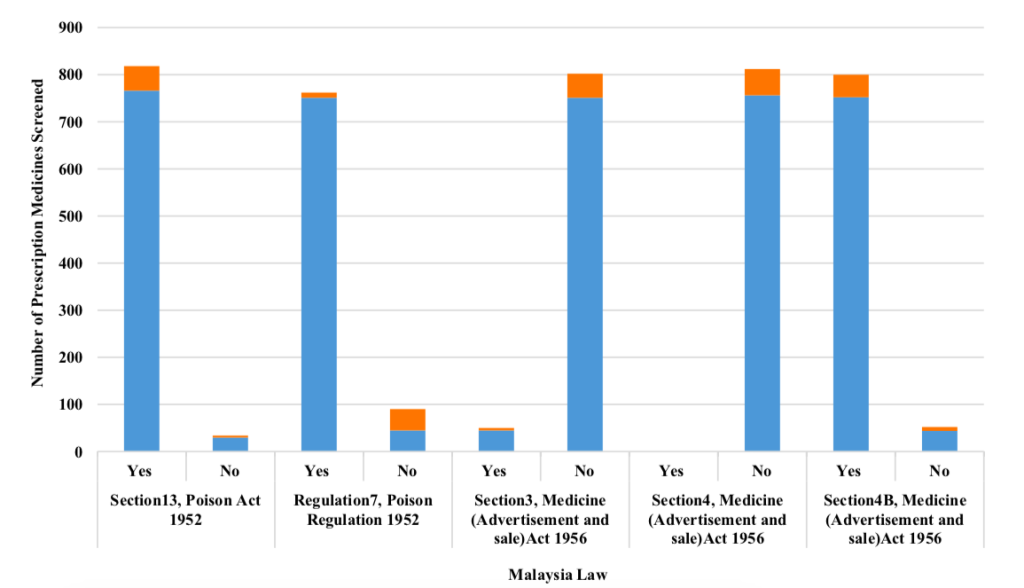
Unregistered prescription medicines were found to be most frequent violations of Section 13(a) of the Poison Act 1952 (Prescription medicines sold by unlicensed personnel), Regulation 7(1)(a) of CDCR 1984 (Prescription medicine sold is not registered with Malaysia’s Health Ministry), and Section 4B MASA 1956 (Advertisements of medicines without approval from the Medicine Advertisements Board) with 766 samples, 751 samples, and 752 samples, respectively (Figure 3). Additionally, Section 3(1) MASA 1956 (Prohibition of advertisements relating to certain diseases listed in MASA 1956) was also violated by 45 samples for unregistered prescription medicines and 5 samples for registered prescription medicines (Figure III). This study also found that no prescription medicines were advertised for abortion in the e-marketplace (specifically: Shopee) which would have violated Section 4 MASA 1956.
Factors Associated with the Patterns of Prescription Medicines Sale through E-Marketplace (specifically: Shopee) in Malaysia.
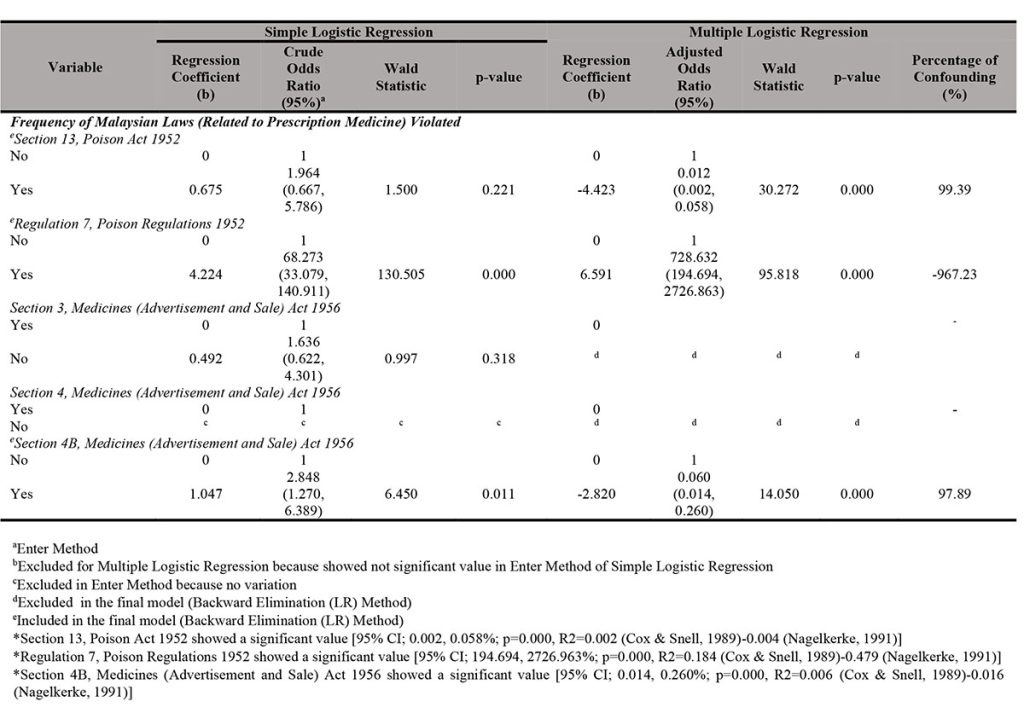
A simple analysis of 852 samples revealed that Regulation 7(1)(a) of CDCR 1984 (which showed 95% CI; 4.224; 68.273%, p = 0.000) and Section 4B MASA 1956 (which showed 95% CI; 1.074; 2.848%, p = 0.011) were the associated factors for registration status of prescription medicines sale through e-marketplace since both p-values were less than 0.05. Conversely, types of prescription medicines were not found to be a factor of association (Table II[a] and Table II[b]).
Multiple logistic regression tests (enter method and backward elimination LR method) presented that Section 13 (a) of the Poison Act 1952 (Adjusted OR: 0.012; 95% CI; 0.002, 0.058%; p = 0.000), Regulation 7(1)(a) of CDCR 1984 (Adjusted OR: 728.63; 95% CI; 194.69, 2726.96%; p = 0.000) and Section 4B MASA 1956 (Adjusted OR: 0.060; 95% CI; 0.014, 0.260%; p = 0.000) were the possible associated factors of registration status of prescription medicine sale through e-marketplace since the p-values were less than 0.05. From the results, it can be interpreted that sellers who violated Regulation 7(1)(a) of CDCR 1984 seemed to have higher chance to sell unregistered medicines compared to those who violated Section 13(a) of the Poison Act 1952 and Section 4B MASA 1956. Section 13(a) of the Poison Act 1952, Regulation 7(1)(a) of CDCR 1984 and Section 4B MASA 1956 were also identified as the potential confounders in this study. Types of prescription medicines sale, Section 3(1) MASA 1956, and Section 4 MASA 1956 were not the significant factors that affected the unregistered products sold on Shopee.
DISCUSSION
As shown in the study, about 93.4% of prescription medicines advertised were unregistered products and the details of the types of prescription medicines involved (as in Table I) alarmed the researchers. This finding shows that the public is widely exposed to the possibility of buying unregistered products and taking medicines without going through the proper channel, which can cause harm to the users. Although this study found that 6.6% of these prescription medicines were registered products, these prescription medicines cannot be advertised at all (unless exempted as per Appendix 1 of Guideline on Advertising of Medicines and Medicinal Products to the General Public).
The current situation in Malaysia is deteriorating as the study shows that most of the sellers were unauthorised laypeople (multiple logistic regression test presented that Section 13(a) of
the Poison Act 1952 showed a significant p-value) and were advertising in a manner that violated MASA 1956 (multiple logistic regression test showed that Section 4B of MASA 1956 showed a significant p-value).
Thus, the recent situation has alerted Malaysia’s enforcement agencies to take proactive and effective actions for controlling and combating this issue to avoid worsening this current condition. The current practice by the government of Malaysia includes instructing the e-marketplace to drop the advertisements that are found to be violating those acts and regulations. This is a good action taken by the Malaysian government. However, some other practical and robust actions may be needed.
The Medicines (Advertisement and Sale) Act 1956 and Regulations only provide the interpretation of advertisement in Section 2 as ‘advertisement includes any notice, circular, report, commentary, pamphlet, label, wrapper or other document, and any announcement made orally or by any means of producing or transmitting light or sound’. Meanwhile, Section 13 of the Poison Act 1952 only emphasises that the possession for sale of poison and sale of poison in contravention of this Act is an offence. Section 16 of the Poison Act mentions that the sale of poisons by retail must be on the premises that had been licensed. Section 18 (1)(b) of the Poison Act 1952 states the restriction on the sale of Part I poisons generally by retail where the sale must be affected by or under the immediate personal supervision of a licensed pharmacist. Section 19 of the Poison Act describes the supply of poisons for the purpose of treatment by professionals. However, these laws and regulations do not control the online prescription medicine advertisement. In short, there is no provision obstructing the advertisement of prescription medicines through online platforms in Malaysia.
Thus, future solutions through the development of internet regulatory technologies and the resolution of jurisdictional issues must be supported by international co-operation [3]. Fraudulent and dangerous practices such as the supply of prescription-only medicines without a medical consultation are an important issue that must be handled by international co-operation [3]. Uniform laws enacted or agreed by each country could result in international treaties or conventions, although reaching consensus is both lengthy and difficult [3]. Internet service providers must be directed to banish sites that do not adhere to standards and the development of a ‘cybercourt’ jurisdiction should be considered [3].
As medications have an impact on one’s health, it is important that policy makers and authorities are alert [1]. Fittler et al. (2018) found that the efforts against online medication purchase are futile as this is a general tendency throughout society [1].
However, the study also proposed that it is essential to develop a stronger control over online pharmacies and provide education regarding responsible internet purchase behaviour [1].
A study in 2011 by Orizio et al. suggested that there are two levels of approaches that could be adopted to enhance the benefits and minimise the risks of online pharmacies [4]. The first level should focus on policy, with laws regulating the phenomenon at an international level [4]. Moreover, Bessell et al. (2002) suggested that strategies to improve the quality of e-pharmacies include independent third-party regulation of providers, evaluation and enforcement of sanctions in cases of dissemination of fraudulent or harmful information and practices, self-regulation, and consumer education [3]. The USA, UK, Canada, Australia and New Zealand have introduced self-regulatory internet pharmacy practice standards, however not all e-pharmacies operating within these countries comply with these standards [3]. Fittler et al. (2018) suggested that the mail order of medicines performed via the internet must be in accordance with licensing requirements and the quality assurance standards of the country of operation [1].
The New Zealand Ministry of Health introduced new legislation in late 2002 which required a prescription to be written by a New Zealand doctor before medicines are supplied overseas [3]. The legal but unethical practice before this was prescription medicines being supplied overseas without any prescription [3]. This new legislation was made after the discovery of large amounts of sildenafil, orlistat, and finasteride being dispatched from New Zealand to the United State and United Kingdom [3].
The second level needs to focus on individuals, with the aim of increasing health literacy (which is required for making appropriate health choices, recognising risks and making the most of the multitude of opportunities offered by the world of medicine 2.0) [4]. Consumer education and awareness programmes must accompany self-regulatory initiatives in order to successfully direct consumers to e-pharmacies accredited by independent third-party authorities [3]. Consumers must be educated about the benefits and risks of buying medicines online instead of being influenced by the online promotion and availability of medicines with no prescription required [3]. Moreover, public education on the safe use of internet pharmacies can be generated by improving patient-provider communication, promotion, and education campaigns [1].
In summary, in order to achieve positive change, international cooperation, surveillance and a partnership approach between all stakeholders where evaluation, regulatory and educational resources can be shared are necessary [3]. The challenge is to discourage fraudulent and misleading websites but permit the development of innovative, ethical e-pharmacy services [3]. Therefore, patient-provider communication should be improved and promotion campaigns are needed to inform the public about the safe use of internet pharmacies, since these initiatives can directly reduce patient safety risks [1]. A study in 2020 found that the most important task for online pharmacies is to improve logistics [6]. The study also suggested that it is better to develop self-built logistics [6].
The discussion on how to best safeguard the global public and ensure the standards of care provided by e-pharmacies is vital across and within countries [3]. The discussion may involve consumers, clinicians including clinical pharmacologists, other health professionals, policy makers, media and lawyers [3]. Additionally, health providers which include general practitioners, pharmacists and nurses may help patients navigate potential internet purchases as a prevention of medication incidents generated using unapproved and illegitimate online pharmacies [1].
This study involved searching for prescription medicines through keyword searches (i.e., via Shopee online platform) of prescription medicine types (such as antifungals, antibiotics and dermatological agents) and medical claims (such as nausea, vomiting, inflammation, abortion and fertility), thus there may have been bias while selecting the advertisements to be screened. Error in reporting may occur but fewer in quantity because the reporting underwent critical screening and checking by three (3) levels of professionals. This study demonstrated variance for the findings of frequently violated Malaysian laws related to prescription medicines by sellers on Shopee; Section 13(a) of the Poison Act 1952 (Prescription medicines sold by unlicensed personnel) as well as Section 4B of MASA 1956 (Advertisements of medicines without approval from Medicine Advertisements Board). Practically, it is well understood that prescribing medicines on Shopee is not a legal act unless exempted as per Appendix 1 of Guideline on Advertising of Medicines and Medicinal Products to the General Public, thus violating Section 13(a) of the Poison Act 1952. However, the level of understanding of this issue by the advertisement screening personnel (enforcement officers) may vary because the sellers’ authorisation was not verified during this study, which may be reflected in the findings. Furthermore, some of the advertisement screening personnel (enforcement officers) were not aware that every advertisement which violated Section 3 of MASA 1956 would also violate Section 4B of MASA 1956, thus these may be reflected in the results.
CONCLUSION
Almost all prescription medicines (796 from 852 approved samples; 93.4%) were found not registered with the Drug Control Authority (DCA), Health Ministry of Malaysia. This study found that the types of prescription medicines sold through e-marketplace (specifically: Shopee) in Malaysia from January till June 2020 varied. Hormones group (533 samples; 62.6%) showed the highest value while sex hormones (i.e., steroid compounds with androgenic substances; commonly known as anabolic steroid) were higher (497 samples; 58.3%) than other types of prescription medicines. Meanwhile, miscellaneous group (80 samples; 9.4%), antibiotics (41 samples; 4.8%) as well as antineoplastic agents and immunosuppressants (40 samples; 4.7%) were also found to be sold at relatively high numbers. Additionally, clomiphene (known as fertility drug with 19 samples; 2.2%) and sibutramine (known as slimming pill with 15 samples; 1.8%) were found to be frequently sold through the e-marketplace (specifically: Shopee). Although this study found that there were no prescription medicines advertised for abortion, which violates Section 4 MASA 1956 in the e-marketplace (specifically: Shopee), there were abortion pills being sold with 3 samples (0.4%). Section 13 (a) of the Poison Act 1952 was found to be the among the most frequently violated laws with 766 samples of unregistered prescription medicines breaching the section, Regulation 7(1) (a) of CDCR 1984 with 751 samples and Section 4B MASA 1956 with 752 samples. Section 3(1) MASA 1956 was also violated by 45 samples for unregistered prescription medicines and 5 samples for registered prescription medicines. Section 13(a) of the Poison Act 1952, Regulation 7(1)(a) of CDCR 1984 and Section 4B of MASA 1956 were the possible associated factors of registration status of prescription medicines sold through e-marketplace and were the identified potential confounders. The findings in this study may provide enforcement agencies in Malaysia as well as any non-government organisation with ideas for improving the current practice in order to curb the freely available illegal prescription medicines sale through e-marketplace (specifically: Shopee) i.e. prescription medicines sold without supervision of professionals such as doctors and pharmacists.
RECOMMENDATION
The government of Malaysia’s enforcement agencies should focus on anabolic steroids when screening prescription medicine advertisements through Shopee since this drug was found to be advertised with the highest frequency. Other than that, special guidelines or regulations that contain stricter policies to be followed by the platform provider of online sale of fertility drug, slimming pill and abortion pill need to be created since they were also found to be advertised through Shopee without supervision of professionals. The present practice of removing problematic advertisements seem to be insufficient. Meanwhile, the special guidelines or regulations should also emphasise the advertisements of medicines without approval from the Medicine Advertisements Board (MAB) since this study found that Section 4B MASA 1956 was the Malaysian law related to prescription medicines being violated with the second highest frequency. For example, since 2002, the Medical Council of New Zealand’s guidelines required that the doctor and consumer must have met on at least one occasion before prescription medicines can be sold online [3]. Other proactive measures that can be done include increasing penalties in the existing guidelines, regulations, or policies. Obligation to display and disclose sellers’ locations and address as well as hand phone numbers is also recommended. The permissions for the advertisements of prescription medicines should be reviewed and the benefits as well as disadvantages must be studied clearly. Moreover, there is a need to control the platform providers by the Malaysian Communications and Multimedia Commission (MCMC), since most of the prescription medicines sold through Shopee were unregistered and Regulation 7(1)(a) of CDCR 1984 was found to be the third most frequently violated Malaysian law related to prescription medicines. Also, Regulation 7(1)(a) of CDCR 1984 which emphasises that the prescription medicines being sold are not registered with the Malaysian Health Ministry was found to be a possible associated factor of registration status of prescription medicines sold through e-marketplace as well as a potential confounder. Meanwhile, Section 13(a) of the Poison Act 1952 which regulates prescription medicines sold by unlicensed personnel was also found to be a possible associated factor and a potential confounder.
Thus, some proactive actions should be regulated through a collaboration of government and non-government agencies. For example, on 15th October 2020, the Malaysian Pharmaceutical Society (MPS) signed a memorandum of understanding (MOU) with Shopee [11]. This memorandum includes MPS verification of pharmacy-related accounts on Shopee [11]. All accounts are to fulfill these two criteria in order to be verified: (i) be a pharmacy with poison license A, and (ii) have a physical store. Once approved, these accounts will be able to receive an MPS-verified badge on their shop fronts [11]. This is a great way for the public to know if purchased products are genuine, safe and registered [11]. However, products sold are limited to supplementary products, traditional products and over-the-counter (OTC) products [11]. Poisons, or in other terms, pharmacy-only and pharmacist-only medication can only be purchased in-stores [11]. However, the collaboration of the internet providers, MPS and Malaysia’s government enforcement agency will be beneficial through the creation of more effective guidelines, regulations or policies in ensuring only authorised sellers with registered products are allowed to advertise online. Self-screening medicine advertisements by the Malaysian government enforcement agency are suggested rather than auto-approved medicine advertisements by the platform providers. Bessel et al. (2002) recommended other actions such as sending warning letters to firms selling unapproved new drugs online and issuing ‘import alerts’ to online sellers of foreign pharmaceuticals as being more cost-effective and timelier [3]. Finally, the suggested education and awareness programmes should be conducted not only for the public but also the platform providers.
ACKNOWLEDGEMENT
The authors would like to thank the Director General of Health Malaysia for the permission to publish this article. This research did not include human subjects. This research has been granted approval by the Malaysia Research Ethics Committee.
CONFLICT OF INTEREST
The authors declare no conflict of interest. This case report did not receive any specific grant from funding agencies in the public, commercial or not-for-profit sectors.
REFERENCE
- Fittler A, Vida RG, Káplár M, Botz L. Consumers turning to the internet pharmacy market: Cross-sectional study on the frequency and attitudes of Hungarian patients purchasing medications online. J Med Internet Res. 2018 Aug 22;20(8):e11115. https://www.jmir.org/2018/8/e11115/
- Aris NA. Ministry tracks thousands of online ads for illicit medicines. 2019 Apr 6 [cited 2020 Apr 6]. Available from: https://www.freemalaysiatoday.com/category/nation/2019/04/06/ministry-tracks-thousands-of-online-ads-for-illicit-medicines/
- Bessell TL, Silagy CA, Anderson JN, Hiller JE, Sansom LN. Quality of global e-pharmacies: can we safeguard consumers?. Eur J Clin Pharmacol. 2002;58(9):567-72. https://doi.org/10.1007/s00228-002-0519-5
- Orizio G, Merla A, Schulz PJ, Gelatti U. Quality of online pharmacies and websites selling prescription drugs: A systematic review. J Med Internet Res. 2011;13(3):e74. https://www.jmir.org/2011/3/e74/
- Mackey TK, Nayyar G. Digital danger: A review of the global public health, patient safety and cybersecurity threats posed by illicit online pharmacies. Br Med Bull. 2016 Jun;118(1):110-26. https://doi.org/10.1093/bmb/ldw016
- Liu J, Zhou Y, Jiang X, Zhang W. Consumers’ satisfaction factors mining and sentiment analysis of B2C online pharmacy reviews. BMC Med Inform Decis Mak. 2020;20(1):1-13. https://doi.org/10.1186/s12911-020-01214-x
- Wang B, Liu J, Kesselheim AS. Variations in time of market exclusivity among top-selling prescription drugs in the United States. JAMA Intern Med. 2015;175(4):635-7. https://doi.org/10.1001/jamainternmed.2014.7968
- Isa K, Foo J, Palpanadan ST, Isa F. Online shopping trend and trust during COVID-19 Movement Control Order. Solid State Technol. 2020;63(1s):1796-803. https://solidstatetechnology.us/index.php/JSST/issue/view/45
- LaMorte WW, Sullivam L. Confounding and effect measure modification [Internet]. Boston University School of Public Health. United States of America: Department of Epidemiology. 2018. https://sphweb.bumc.bu.edu/otlt/mph-modules/bs/bs704-ep713_confounding-em/bs704-ep713_confounding-em_print.html
- Wiedmann KP, Hennigs N, Pankalla L, Kassubek M, Seegebarth B, Reeh MO. Online distribution of pharmaceuticals: investigating relations of consumers’ value perception, online shopping attitudes and behaviour in an e-pharmacy context. J Cust Behav. 2010;9(2):175-99. https://doi.org/10.1362/147539210X511362
- Tan SXY. MOU between MPS x Shopee [Internet]. MPS Young Pharmacists Chapter. 2020 Nov 4. https://mpsypc.com.my/publications/mou-between-mps-x-shopee/
- Kaur H, Kunasegaran M, Singh J, Salome S, Sandhu SK. Impact of the First Phase of Movement Control Order during the COVID-19 pandemic in Malaysia on purchasing behavior of Malaysian Consumers. Horizon J Hum Soc Sci. 2020;2(S):131-144. https://horizon-jhssr.com/view-issue.php?id=28
- Wang B, Liu J, Kesselheim AS. Variations in time of market exclusivity among top-selling prescription drugs in the United States. JAMA Intern Med. 2015;175(4):635-7. https://doi.org/10.1001/jamainternmed.2014.7968
- Chaturvedi AK, Singh UK, Kumar A. Online pharmacy: An e-strategy for medication. Int J Pharm Front Res. 2011;1(1): 146-58. https://www.researchgate.net/publication/237201481_ONLINE_PHARMACY_AN_E-STRATEGY_FOR_MEDICATION
- Alwon BM, Solomon G, Hussain F, Wright DJ. A detailed analysis of online pharmacy characteristics to inform safe usage by patients. Int J Clin Pharm. 2015;37(1):148-58. https://doi.org/10.1007/s11096-014-0056-1
- Kuzma J. Web vulnerability study of online pharmacy sites. Inform Health Soc Care. 2011;36(1):20-34. https://doi.org/10.3109/17538157.2010.520418
- Lee JL, Choudhry NK, Wu AW, Matlin OS, Brennan TA, Shrank WH. Patient use of email, Facebook, and physician websites to communicate with physicians: A national online survey of retail pharmacy users. J Gen Intern Med. 2016;31(1):45-51. https://doi.org/10.1007/s11606-015-3374-7
Please cite this article as:
Marina Pilus, Malar Vily Velisamy, Nurain Suleiman, Noor Azline Ali, Farizul Mohd Zain and Augustine Abraham Alphonsoes, Patterns of Prescription Medicines Sale Through E-Marketplace in Malaysia and Associating Factors. Malaysian Journal of Pharmacy (MJP). 2021;2(7):85-97. https://mjpharm.org/patterns-of-prescription-medicines-sale-through-e-marketplace-in-malaysia-and-associating-factors/