Abstract
Introduction: Reports of baclofen toxicity in patients with severe renal impairment have raised concerns regarding prescribing practice of the drug in the outpatient department. The aim of this study is to determine the incidence of inappropriate prescribing of baclofen and its prescribing pattern. Method: This was a retrospective, observational study of prescriptions from July to December 2014 in an outpatient clinic of a public hospital in Malaysia. All prescriptions containing baclofen were selected and records of these patients were retrieved and reviewed. Where available, results of serum creatinine were also collected. Appropriateness of baclofen use was determined by 3 independent doctors based on labelled indication of baclofen. Result: Out of 65,922 prescriptions screened, 691 (1.1%) prescriptions which contained baclofen and whose records could be retrieved were included in the analysis. Most of the prescriptions were for pain (78.2%) and contained at least one pain medication. Baclofen was prescribed concomitantly with NSAIDs, antihypertensives and oral hyperglycaemic agents in 535 (77.4%), 49 (7.1%) and 25 (3.6%) cases respectively. Two patients had kidney failure. The overall proportion of inappropriate use of baclofen was high (n = 641, 92.7%). Although the trend of baclofen use reduced drastically after September 2014, possibly due to stricter prescribing practices that was enforced in the department, the proportion of its inappropriate use remained high. Conclusion: Inappropriate use of baclofen is prevalent. Rigorous interventions such as trainings should be carried out to avoid future preventable overdoses or toxicities.
Introduction
In 2014, 5 cases of baclofen toxicity had been reported in patients with severe renal impairment in a public hospital in Malaysia. In the aforementioned report, all five patients presented with an acute onset of altered sensorium within 24 – 48 hours of ingesting baclofen with total doses ranging from 20 – 40 mg. They had been prescribed baclofen for muscle ache and backache from the outpatient department (OPD).
Baclofen is an antispasmodic medication which a structural analogue of γ-aminobutyric acid (GABA) that acts at the spinal cord level by inhibiting both monosynaptic and polysynaptic reflexes [1], resulting the relief of muscle spasticity. It has been shown to be effective in the treatment of muscle spasticity due to multiple sclerosis and spinal lesions of traumatic, infectious, degenerative, neoplastic and unknown origin that show symptoms of skeletal hypertonus, and spastic and dyssynergic bladder dysfunction [2]. However, it is not recommended for patients with Parkinson’s disease or spasticity caused by strokes, cerebral palsy or rheumatoid disorders. Its lipophilicity and penetration into the cerebrospinal fluid are responsible for its CNS side effects such as sedation, fatigue, dizziness, lowering of the seizure threshold and cognitive dysfunction. Interactions and adverse reactions may occur when baclofen is taken concomitantly with other drugs such as tricyclic antidepressants, MOA inhibitors and levodopa.
Because baclofen is predominantly excreted unchanged by the kidney, its clearance is significantly decreased, in patients with severely impaired renal function, thus prolonging its half-life. In fact, previous studies have reported altered consciousness, seizures, hallucinations, hypothermia, bradycardia, hypertension, abdominal pain [3], progressive confusion, generalized decrease in muscle tone [4], encephalopathy, ataxia and dystonia [5] in renal failure patients with baclofen intoxication. It is therefore suggested that baclofen should be prescribed only when it is clinically indicated whereby when the expected benefit outweighs the potential risks, it is to be prescribed at a lower dose and with caution [2].
The appropriateness of drug use is crucial in ensuring and ascertaining the reduction of patient morbidity and mortality, improving quality of life and in containing drug and healthcare expenditure. Unfortunately, despite its importance, the irrational use of drugs is still a global problem and few countries have taken sufficient actions to correct the situation [6]. The aim of this study is to determine the incidence proportion of baclofen prescribing and its appropriateness in a tertiary hospital in Malaysia and to describe its prescribing pattern.
Method
Study Design
This report involves a retrospective, observational study to determine the pattern and appropriateness of baclofen prescribing in an outpatient clinic of a tertiary public hospital in Malaysia. Prescriptions from the clinic from July to December 2014 were screened, from which, all prescriptions containing baclofen were selected. Consecutively, records of these patients were retrieved and reviewed. Patient demographics, symptoms, diagnosis, baclofen dose and duration, concomitant medications, comorbidities, medical history and prescriber category were recorded. Additionally, results of serum creatinine were also retrieved if available. Prescriptions with missing patient records were excluded. The appropriateness of baclofen use was determined as per labelled indication and in situations when the diagnosis was not indicated, or where there is doubt, the appropriateness of its use based on the medical records, was decided by three specialists. This study was approved by the Medical Research and Ethics Committee in Malaysia (KKM / NIHSEC / P15 – 589). There were no potential competing interests involving any investigators in this study.
Statistical analysis
Continuous variables were summarised as means and standard deviations if they were normally distributed, or as medians and interquartile ranges if they were non-normally distributed. Categorical variables were summarised as frequencies and percentages. The incidence proportion of baclofen prescribing was estimated from the total prescriptions issued from the clinic throughout the six months of the study, while the incidence proportion of inappropriate use of baclofen was estimated from the total prescriptions with baclofen over the same period of time. All statistical analyses were performed with STATA 11 (StataCorp, College Station, TX).
Result
We screened 65,922 prescriptions from the outpatient department from July to December of 2014. From these prescriptions, 846 prescriptions contained baclofen, thereby resulting in an incidence proportion of baclofen prescribing of 1.3% over the period of six months. Of these prescriptions, 691 (81.7%) clinical records were retrieved and included in the analysis. All prescriptions were ordered by medical doctors, and none were prescribed by medical assistants.
The main demographic profile of the patients with the relevant prescriptions is shown in Table I. There were 104 (15.1%) prescriptions for elderly patients aged 65 years and above and 22 (3.2%) prescriptions for children less than 18 years old. The most frequent comorbidities recorded were hypertension (12.2%), diabetes mellitus (8.3%), dyslipidaemia (4.6%), osteoarthritis (3.8%) and gastritis (2.7%).

Based on demographic profile matching, 123 (17.8%) prescriptions were from return patients who collected their prescriptions more than once within the study period. Another 27 prescriptions had missing birthdates, and thus could not be matched. The repeat prescriptions consist of 50 patients who returned once, 6 patients who returned twice and one patient who returned 4 times.
Table II illustrates the dose, frequency and duration of baclofen prescribed. The dose of baclofen ranged from 10 mg to 60 mg per day, with a mean daily dose of baclofen of 25.9mg ranging from 10mg to 60mg per day. The number of medications per prescription ranged from 2 to 15 with a mean of 5.4 (1.77) medications per prescription.
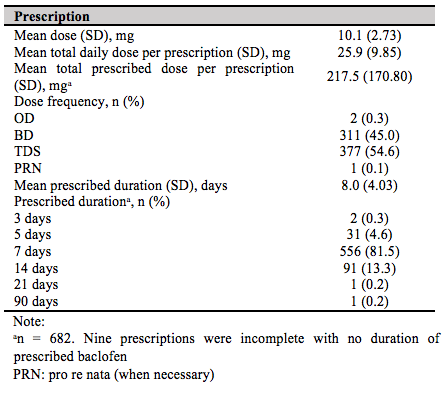
Table III shows the indications for the prescription of baclofen. It is suggested that baclofen had mostly been prescribed for pain (78.2%), whereby the majority of the pain symptoms were related to musculoskeletal pain (91.1%). Of these prescriptions for musculoskeletal pain, 92 prescriptions (18.7%) were related to traumatic or soft tissue injuries, while the remaining 400 (81.3%) were related to non-traumatic events such as joint and tendon pain as well as back pain.
The majority of the prescriptions contained at least one pain medication. We found 623 (90.2%) prescriptions with at least one oral pain medication, 531 (76.8%) with topical methyl salicylate ointment and 29 (4.2%) with intramuscular diclofenac sodium. One prescription contained two concurrent oral NSAIDs. In total, baclofen was prescribed concomitantly with NSAIDs in 535 (77.4%) prescriptions.
Almost one-fourth (165; 23.9%) of the records had information on serum creatinine. Using the Modification of Diet in Renal Disease Study (MDRD) equation for estimated glomerular filtration rate (eGFR), we found 71 (43%) prescriptions whereby baclofen was prescribed for patients who had eGFR below 90 mL / min / 1.73m2, out of which two Stage 4 chronic kidney disease (CKD) (Table I) case were identified. Two other prescriptions were prescribed for patients with kidney failure although, no serum creatinine information was available in the records of these two patients.
Baclofen was prescribed in the same prescription together with antihypertensives and/or diuretics 49 times and with oral hyperglycaemic agents (OHA) 25 times. The mean total daily dose of baclofen in prescriptions with concomitant antihypertensive and/or diuretic medications and concomitant

OHAs were 24.0 (SD 8.35) mg and 22.4 (SD 10.52) mg, respectively. The minimum and maximum daily baclofen doses in both these groups were 10 mg and 60 mg respectively.
Table IV shows the breakdown number of prescriptions containing baclofen by month and the appropriateness of its use. Aside from the month of August, where the number of prescribed baclofen was the highest, the general baclofen prescribing trend had been decreasing. The decrease was most prominent in the months of October, November, and December of 2014.
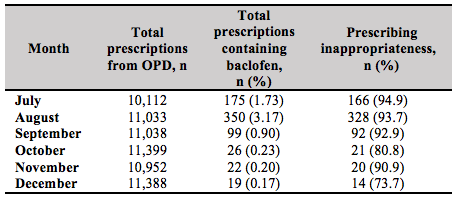
The inappropriate use of baclofen was high with an incidence proportion of inappropriate baclofen of 92.8% over the six months study. Prescriptions and records with neither symptom nor diagnosis were considered inappropriately prescribed. Some of the common symptoms and diagnoses for these inappropriately prescribed prescriptions were undefined back pain (9.8%), undefined muscle pain (5.9%), shoulder pain (4.7%), fall (3.2%) and headache (2.6%). Additionally, the proportion of inappropriate use did not change much throughout the six months.
Discussion
Baclofen toxicity in severely impaired renal function patients is relatively rare despite being well-acknowledged for a long time. In fact, several cases of baclofen toxicity IN GENERAL have been reported, dating all the way back to 1976 have reported baclofen overdoses [7][8]. Elsewhere, Chen KS et al. reported nine cases of baclofen toxicity in such patients in a hospital in Taiwan from 1991 to 1995 [3]. More recently, there were another two reported cases of baclofen toxicity in End-Stage Renal Disease (ESRD) in the United States [4][5], where one of the two patients was prescribed baclofen for muscle spasms while the other patient was prescribed the drug for back pain. For the case of both patients, however, baclofen use for muscle aches, backaches and general muscle spasms could have been potentially inappropriate.
For the case of our study, we had found a sizable number of prescriptions for baclofen with no records of diagnoses or symptoms indicating the use of the drug. Furthermore, some of those prescriptions were only indicated for the refill of baclofen with no documentation of its indication. Such critically incomplete records are a cause for concern. Meanwhile, the remaining records with documented symptoms and diagnoses had little-to-no elaboration of the cause or origin of the complaint.
In most cases in our study, baclofen was found to not be the first-line choice of drug for its corresponding indication, and further justification of its use can only be attained by further elucidating information gathered from the records. Evidence-based guidelines would suggest that its use is considered possibly appropriate if it was prescribed for muscle stiffness or prolapsed intervertebral disc. Besides that, its use is also deemed possibly appropriate in prescriptions for muscle spasms in accordance with the NICE guideline on the treatment of spasticity for those aged under 19 years old [9], whereby the guidelines recommended baclofen for muscle spasms caused by spasticity. In addition to that, some studies have reported on the effectiveness of baclofen in treating hemifacial spasms [10][11]. In spite of those guidelines and reports, we could not preclude its appropriateness due to inadequate information on the diagnoses of muscle stiffness, spasms and hemispasm in our cohort.
Despite how the diagnoses of several cases were left dubious, we were able to determine definitively that baclofen was inappropriately prescribed in more than 90% of the prescriptions. These prescriptions involved the dispensing of baclofen for gastritis and abdominal pain, headaches, pain or cramps in the limbs, upper respiratory tract infection as well as chest pain, all of which are indications for baclofen use that could not be justified.
When baclofen is used together with drugs that could impact renal function such as NSAIDs, its excretion could be significantly reduced and may lead to baclofen toxicity. Of the 691 prescriptions, 77.4% contained at least one concomitant NSAIDs prescribed but only 23.9% of these prescriptions had records of creatinine. In fact, creatinine tests are usually not indicated in the premise for patients in the OPD unless there is reason to suspect renal impairment. Of the cases with available serum creatinine information, it is found that 43% of patients had kidney function below the normal range based on the Kidney Disease Outcomes Quality Initiative (KDOQI) CKD staging (with eGFR below 90 mL / min / 1.73m2). It can be surmised that for such cases, doctors prescribing baclofen who had been uninformed of a patient’s comorbid kidney disease may cause potential baclofen accumulation, leading to toxicity. In support of this, it is found that most cases of baclofen toxicities in the literature were in patients with ESRD [3][4][5], which also include five locally reported toxicity cases. Therefore, it is important to prospectively ensure that renal function is not impaired before prescribing baclofen and to avoid using it in end-stage kidney disease.
Furthermore, the implications of polypharmacy and drug-disease interactions in baclofen use is also an important consideration. Dhabali et al. reported that higher numbers of drug-drug interactions were seen in older patients and in those with multiple prescribed medications [12]. In our sample, most of the prescriptions contained between four to six drugs, increasing the risk of drug interactions. Besides that, we have also found several cases where baclofen was prescribed in patients with comorbid diabetes mellitus and hypertension. These cases deserve attention because baclofen can increase blood glucose concentrations and concomitant treatment with antihypertensives can enhance the reduction in blood pressure. Nevertheless, assessment and monitoring for side effects could, unfortunately, not be done as they were outpatients. To exacerbate the occurrence of drug-disease and drug-drug interactions, many of the hospitals in Malaysia have yet to incorporate an integrated electronic medical record system, and patients may have comorbid conditions with corresponding medications that are prescribed elsewhere unbeknownst to the OPD. Thus, doctors will have to be more careful when prescribing to prevent such medication interactions and errors.
Incorrect dosing for baclofen in renal failure patients, oftentimes leading to an overdose, is a serious issue that cannot be overlooked. A study carried out on medication errors reported to the National Medication Error Reporting System in Malaysia found that more than 75% of the errors were attributed to the prescribing stage. Incorrect drug dosage was reported to be the most common error [13]. A review paper on medication errors in Southeast Asia by Salmasi et al. also revealed incorrect dosing to be the most frequently reported type of prescribing error [14]. A survey conducted on senior practising hospital pharmacists in West Malaysia found that 60% of the participants agreed that prescribing errors among doctors were common [15]. Another study carried out in the US also found most (70%) medication errors originated from the prescribing stage and incorrect dose and drug selection were the two most commonly reported errors [16].
Studies have shown that several factors may contribute to such inappropriate prescribing or prescribing errors. Shortage of staff, overworked staff, distractions, prescriber level of knowledge or lack of training, prescriber conflicts of interest, previous prescribing experience or practice as well as failure to comply with clinical guidelines and patient demand can all lead to errors [14][17][18]. On average, the OPD of this hospital sees close to 800 patients a day, which is an exorbitant amount that can be attributed to the fact that it is inexpensive to seek treatment from public hospitals in Malaysia. The number of staff is not commensurate with the high number of patients, leading to overworked and distracted staff, and possibly contributing to poor documentation practices in the department.
Stricter prescribing practices for baclofen were enforced in the OPD in September 2014 after five cases of baclofen toxicity in chronic kidney patients were reported. Consequently, the effect of this newfound awareness was reflected in the drastically reduced number of prescribed baclofen beginning in September 2014. Additionally, interventions to assuage this issue, including continuous professional education sessions and mandatory validation by the head of departments for every prescribed baclofen were implemented. This need for validation may deter junior doctors from prescribing the drug which could explain the reduction in its prescription. Despite these actions, however, the proportion of its inappropriate use did not change much, a fact that may be attributed to the passing down of misinformation, whereby previous malpractice involving baclofen is handed down from one generation of doctors to another.
As of December 2014, we saw a larger drop in inappropriate use, suggesting that the education sessions were taking effect. To supplement these educational endeavours, guidelines and training on guidelines should be introduced and carried out more often to ascertain a more lasting impact. A review by le Grand et al. found that standard treatment guidelines in several countries such as Kenya, Fiji and Indonesia have had positive changes in prescribing practices [18]. Unfortunately, this was a short study, and we were not able to determine if the drop persisted in later months. Nevertheless, guidelines and training appear to be promising and can be explored further in future interventions. Proper and more detailed recording should also be enforced to prevent future medication errors due to a lack of medical information.
Conclusion
The proportion of inappropriate use of baclofen was very high. Although the number of baclofen prescribed was significantly reduced after the enforcement of stricter prescribing practices, the same could not be demonstrated in the proportion of its inappropriate use. Therefore, more frequent and rigorous interventions should be enforced to ensure lasting improved prescribing practices to avoid any risk of future preventable overdoses or toxicities.
Acknowledgement
We would like to thank Dr Chan Wei Heng (Department of Orthopaedics, Penang Hospital), Dr Choo Chin Yin (Department of Orthopaedics, Hospital Seberang Jaya), Dr Yoon Chee Kin (Department of Medicine, Penang Hospital) and the staffs in the outpatient department of Penang Hospital for their assistance in the study. We would also like to thank the Director General of Health Malaysia for his permission to publish this article. This study was not supported by any grant or funding.
Conflict of Interest
This study has no conflict of interest. This research did not receive any specific grant from funding agencies in public, commercial or not-for-profit sectors.
Reference
- Novartis Pharmaceuticals Canada Inc. Product Monograph Lioresal (baclofen) 10 and 20 mg tablet [Internet]. [cited 2017 Jan 5]. https://www.ask.novartispharma.ca/download.htm?res=lioresal_scrip_e.pdf&resTitleId=802
- Australian Product Information – Lioresal (Baclofen) Tablets [Internet]. [cited 2017 Jan 5]. http://www.guildlink.com.au/gc/ws/nv/pi.cfm?product=nvplrsor10113
- Chen KS, Bullard MJ, Chien YY, Lee SY. Baclofen toxicity in patients with severely impaired renal function. Ann Pharmacother. 1997 Nov;31(11):1315–20 https://doi.org/10.1177/106002809703101108
- Ijaz M, Tariq H, Kashif M, Marquez JG. Encephalopathy and hypotonia due to baclofen toxicity in a patient with end-stage renal disease. Am J Case Rep. 2015 Apr 20;16:232–5. https://doi.org/10.12659/AJCR.893222
- Porter LM, Merrick SS, Katz KD. Baclofen Toxicity in a Patient with Hemodialysis-Dependent End-Stage Renal Disease. J Emerg Med. 2017 Apr;52(4):e99–100. https://doi.org/10.1016/j.jemermed.2016.09.025
- The World Medicines Situation Report [Internet]. [cited 2017 Jan 5]. http://www.who.int/medicines/areas/policy/world_medicines_situation/en/
- May CR. Baclofen overdose. Ann Emerg Med. 1983 Mar;12(3):171–3. https://doi.org/10.1016/s0196-0644(83)80562-9
- Paulson GW. Overdose of lioresal. Neurology. 1976 Nov;26(11):1105–6. https://doi.org/10.1212/wnl.26.11.1105
- Spasticity in under 19s: management [Internet]. London: National Institute for Health and Care Excellence (UK); 2016 [cited 2021 Nov 17]. (National Institute for Health and Care Excellence: Clinical Guidelines). http://www.ncbi.nlm.nih.gov/books/NBK554883/
- Sandyk R, Gillman MA. Baclofen in hemifacial spasm. Int J Neurosci. 1987 Apr;33(3–4):261–4. https://doi.org/10.3109/00207458708987410
- Sandyk R. Baclofen in hemifacial spasm. Eur Neurol. 1984;23(3):163–5 https://doi.org/10.1159/000115698
- Dhabali A a. H, Awang R, Zyoud SH. Clinically important drug-drug interactions in primary care. J Clin Pharm Ther. 2012 Aug;37(4):426–30. https://doi.org/10.1111/j.1365-2710.2011.01314.x
- Samsiah A, Othman N, Jamshed S, Hassali MA, Wan-Mohaina WM. Medication errors reported to the National Medication Error Reporting System in Malaysia: a 4-year retrospective review (2009 to 2012). Eur J Clin Pharmacol. 2016 Dec;72(12):1515–24. https://doi.org/10.1007/s00228-016-2126-x
- Salmasi S, Khan TM, Hong YH, Ming LC, Wong TW. Medication Errors in the Southeast Asian Countries: A Systematic Review. PLOS ONE. 2015 Sep 4;10(9):e0136545. https://doi.org/10.1371/journal.pone.0136545
- Rahman AR, Noor AR, Hassan Y. How good are doctors as drug prescribers? Med J Malaysia. 1994 Dec;49(4):364–8. https://pubmed.ncbi.nlm.nih.gov/7674972/
- Kuo GM, Phillips RL, Graham D, Hickner JM. Medication errors reported by US family physicians and their office staff. Qual Saf Health Care. 2008 Aug;17(4):286–90. https://doi.org/10.1136/qshc.2007.024869
- le Grand A, Hogerzeil HV, Haaijer-Ruskamp FM. Intervention research in rational use of drugs: a review. Health Policy Plan. 1999 Jun;14(2):89–102. https://doi.org/10.1093/heapol/14.2.89
- Joshua IB, Passmore PR, Parsons R, Sunderland VB. Appropriateness of prescribing in selected healthcare facilities in Papua New Guinea. Health Policy Plan. 2014 Mar;29(2):257–65. https://doi.org/10.1093/heapol/czt012
Please cite this article as:
Chin Chin Ch’ng, Ru Shing Ng, Hong Chin Wee, Serene Li Ching T’ng and Loke Meng Ong, Baclofen Prescribing Practice and Its Appropriateness in an Outpatient Setting of a Tertiary Hospital. Malaysian Journal of Pharmacy (MJP). 2022;1(8):1-6. https://mjpharm.org/baclofen-prescribing-practice-and-its-appropriateness-in-an-outpatient-setting-of-a-tertiary-hospital/