Abstract
Introduction: The Antimicrobial Stewardship (AMS) program had been advocated to promote the rational use of antibiotic prescribing. However, the outcome of AMS in promoting the judicious use of carbapenem, and thus, minimising resistance, has not been widely studied in Malaysia. Objective: To investigate the types of interventions made by the AMS team, their acceptance, and the impact of such interventions on carbapenem consumption as well as the resistance pattern of carbapenem-resistant Enterobacterales (CRE). Method: This was a retrospective study conducted in adult medical wards of the Kuala Lumpur General Hospital (HKL), whereby data was extracted from the AMS forms of patients and subsequently reviewed by the AMS team from January to December 2016. Result and Discussion: The mean (SD) age of 169 patients included in this study was 59.2 (10.6) years. Ertapenem was the most prescribed carbapenem (44.4%), followed by meropenem (34.3%) and imipenem/cilastatin (21.3%). The study demonstrated that only 32% of carbapenem therapy had been empirically initiated, while, 68 cases (40.2%) were classified as unjustified use. Out of these cases, 39 cases (57%) were recommended to be discontinued, 25 cases (37%) were to be de-escalated and 4 cases (6%) were set for changing/escalation. The acceptance rate was reported to be around 73.5% (50 out of 68 cases). After one year of AMS implementation, carbapenem consumption (as shown by the defined daily dose/1000 inpatient bed-days) reduced by 33.7%. Similarly, a notable decrease in CRE cases (33.3%) was observed following a year of AMS initiation. Conclusion: AMS-guided interventions were able to demonstrate a reduction in carbapenem consumption as well as CRE rates in the medical wards.
Introduction
Antibiotic resistance is a major global health threat, which requires prompt attention [1]. The rapid emergence of antibiotic resistance is attributed both to the overuse or misuse of antibiotics as well as the lack of innovation in the development of new antibiotics in recent years. The intensity of antibiotic consumptions per year is worrying, as previous studies have shown the direct relationship between antibiotic consumption and the emergence of resistance [1]. Therefore, there is a need to monitor the appropriate use of antibiotics, as well as to reduce inappropriate antibiotic prescribing [2].
In Malaysia, healthcare-associated infections caused by multidrug-resistant organisms (MDRO) particularly carbapenem-resistant Enterobacterales (CRE) is on the rise, with a two-fold increase in CRE isolates reported between the years 2013 and 2016 (0.02 to 0.05 / 100 hospital admission) [3]. A similar finding was shown in a report by the Kuala Lumpur General Hospital (HKL), the largest tertiary hospital in the country; with an increasing trend in the number of CRE cases reported in 2015 [4]. The severity of the impact brought by these reports should be taken seriously, as infection due to these organisms are associated with significant morbidity, mortality, and increased healthcare cost [2].
In view of this, the World Health Organisation (WHO) reported in 2014 that there is an urgent need to address the global threat of antibiotic resistance through Antimicrobial Stewardship (AMS) program [1][2]. The first World Antibiotic Awareness Week in 2015 had been launched to increase the awareness of antibiotic resistance and encourage appropriate use through an AMS. As a result, AMS programs had been introduced worldwide as one of the key strategies to rationalise antibiotic prescribing, conserve remaining antibiotics, and most importantly, ensure optimal clinical outcomes for patients [1][2]. In line with global efforts, the Ministry of Health Malaysia (MOH) formulated a local AMS protocol to encourage stewardship activities in all healthcare facilities in the country. The protocol emphasised on ten core strategies for AMS activities, one of which is to formalize regular multidisciplinary stewardships rounds [5].
As a concerted effort with MOH’s AMS initiatives, a multidisciplinary AMS team was formed in 2014 with the intention of commencing stewardship activities in HKL. In 2016, the team consolidated its AMS efforts through the initiation of stewardship rounds focusing on carbapenem cases. Carbapenem had been chosen as the antibiotics of concern since the increasing CRE rate was hypothesized to be attributed to the increased in the imprudent use of carbapenem. In Malaysia, there is limited evidence on AMS in terms of its acceptance and impact. Therefore, it is important to investigate the local acceptance of AMS recommendations as well as the impact of stewardship strategies. The results would prove to be of great significance as it not only provides a view on the effectiveness of the program but would also highlight areas requiring improvement for a more successful and effective stewardship program. Thus, the objectives of this study include the following; to investigate the types of interventions made by the multidisciplinary AMS team, the acceptance rate of interventions and the impact on carbapenem consumption and CRE rate.
Method
Study Design and Setting
The study was a retrospective study performed in adult medical wards in HKL. A retrospective review of patient data from the medical records and recommendations made in the AMS tracking sheet was conducted in HKL from January 2016 to December 2016. The HKL AMS team consisted of infectious disease physicians, clinical pharmacists, microbiologists and infection control nurses who provide clinical expertise on a patient-by-patient basis for carbapenem therapy. Patients initiated on carbapenem were routinely reviewed by the AMS team and the use of carbapenem were classified as either “justified” or “non-justified”. Appropriate recommendations were suggested for the cases reviewed, which were recorded into AMS tracking sheets.
Inclusion and Exclusion Criteria
During the one-year study period, the medical records of all hospitalised adult patients in the medical ward with a carbapenem prescription that had been initiated by a physician and reviewed by the AMS team were included in the study. Cases in which carbapenem was initiated by other wards or facilities were excluded.
Data Collection
Data collection was performed using a standardised data collection form. The baseline characteristics collected by the study investigators included demographic profile and details of infection, whereby demographic data collected include age, gender, and ethnicity, while clinical data collected include the type of carbapenem, indication for initiation, source of infection, types of culture and sensitivity sample as well as the microorganisms isolated. The types of recommendations reported in the AMS tracking sheet were also collected. The impact of AMS was determined by comparing the carbapenem consumption and number of CRE cases a year after the AMS was consolidated as compared to when it had first been initiated; 2015 versus 2016. Carbapenem consumption was recorded based on the antibiotic consumption surveillance report [6][7], and expressed as WHO defined daily doses (DDDs) per 1000 patient-day [8]. The number of CRE cases were obtained from the reports published by the pathology department of the hospital [4][9]. Justified use of carbapenem refers to use of carbapenem when it is indicated, whilst providing appropriate and adequate coverage (optimal dose and duration) for the diagnosis or suspected infection based on previous work, meanwhile non-justified use refers to the opposite [10].
Sample Size and Data Analysis
The minimum sample size to achieve 80% study power and 95% confidence interval was 114. The universal sampling method was applied, whereby all cases that fulfilled the eligibility criteria were included in the study analysis. Data analysis was conducted using IBM® SPSS Statistics for Windows, version 24. For continuous variables, the differences in mean between two groups were compared using the independent sample t-test and reported as means (standard deviation, SD). For categorical variables, either the Chi-squared or the Fisher’s exact test was used, whereby result are reported as proportions (percentage, %). The CRE rates and carbapenem consumption were analysed using the Chi-squared test and t-test respectively. Statistical significance was denoted by p value < 0.05.
Ethics Approval
Ethical approval was obtained Medical Research and Ethic Committee, Ministry of Health Malaysia (NMRR:17-577-34878).
Result
Demographic and Clinical Characteristics
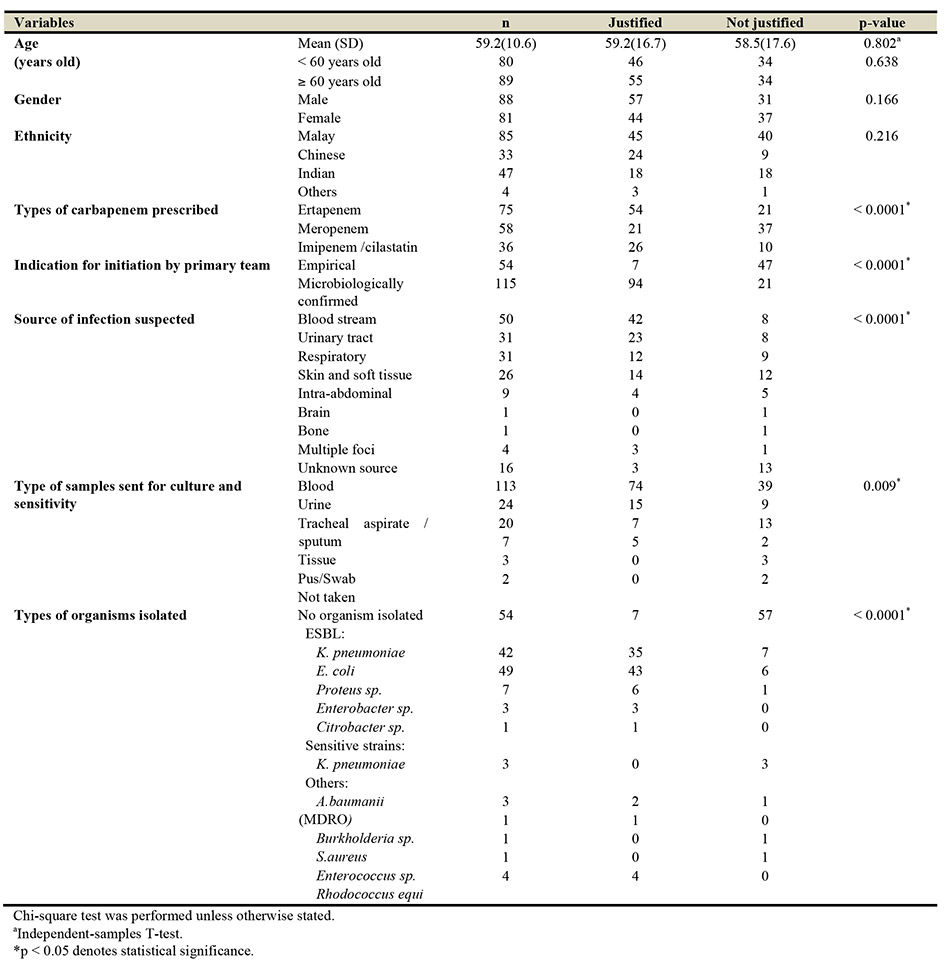
From January to December 2016, 172 AMS cases reviewed by the team. However, only 169 were included in this study based on the inclusion and exclusion criteria. The demographic profile and clinical characteristics of these patients are as described in Table I. The mean (SD) age of patients included in this study was 59.2 (10.6) years, where almost half of them were more than 60 years old. An equal proportion of male and female patients were included in the study. Ertapenem was the most prescribed carbapenem (44.4%), followed by meropenem (34.3%) and imipenem/cilastatin (21.3%). Carbapenems were initiated empirically in 54 out of the 169 cases (32%), where 80% of empirical prescriptions were for meropenem. About 70% (n = 111) of the cultures obtained were sourced from either the blood (n=50), the urinary tract (n = 31), or the respiratory tract (n = 31). Of the 115 microbiologically confirmed cultures, extended-spectrum beta-lactamases (ESBL) producing microorganisms were isolated in 102 (88.7%) of the cases. There were 54 cases (32%), where there were no positive cultures isolated.
Types of AMS Recommendations and Acceptance Rate
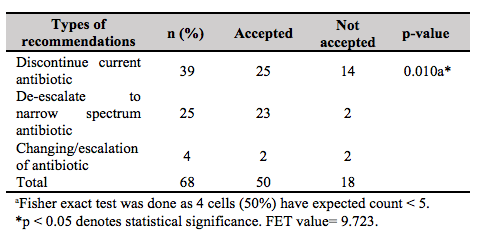
Upon review by the AMS team, it was found that only 101 (59.8%) cases of carbapenem use were justified. Meropenem was the most common carbapenem that was used injudiciously (37 cases, 54.4 %). The types of recommendations given by the AMS team to rectify the non-justified use of antibiotics (68 cases) include discontinuation of therapy (39 cases), de-escalation of therapy (25 cases) and changing/escalation of therapy (4 cases), as shown in Table II. The acceptance rate by the primary team towards these recommendations was 73.5% (n = 50). There was a significant association between the types of recommendations given and its acceptance (p = 0.01). De-escalation was the most accepted recommendation (23 out of 25 cases, 92%), followed by discontinuation of therapy (25 out of 39 cases, 64.1%) and change/escalation of therapy (2 out of 4 cases, 50%). The two cases accepted for escalation of therapy involved skin and soft tissue infection caused by MDRO Acinetobacter baumanii and Enterococcus sp. bacteraemia, whereby colistin and vancomycin had been suggested respectively. The reasons of non-acceptance (n = 18, 40.2%) identified from this study included (I) patients were responding clinically (n = 5, 27.8%), (II) severely ill patients (n = 6, 33.3%), (III) immunocompromised patients (n = 3, 16.7%) and (IV) intention for short duration of carbapenem prescription (one week) (n = 4, 22.2%).
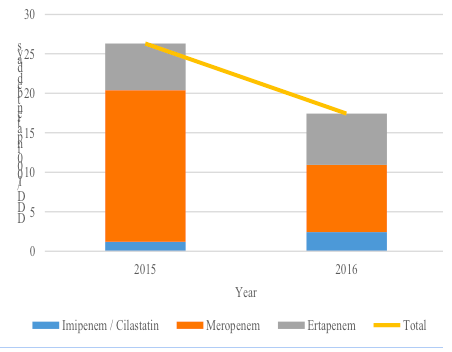
Impact of AMS on Carbapenem Consumption and CRE Cases
The impact on carbapenem consumption after a year of AMS implementation is as shown in Figure I, expressed as DDD/1000 inpatient bed-days for year 2015 and 2016 [6][7]. Overall, there was a 33.7% reduction in carbapenem consumption (p < 0.001) from 26.3 DDD / 1000 inpatient bed-days in the year 2015 to 17.4 DDD / 1000 inpatient bed-days in the year 2016. There was a 55.7% decline in meropenem use from 2015 to 2016; 19.20 DDD / 1000 inpatient bed-days to 8.51 DDD/1000 inpatient bed-days, respectively. However, the consumption for ertapenem and imipenem / cilastatin escalated
between 2015 and 2016 with a 2-fold increase for imipenem / cilastatin (1.20 to 2.43 DDD / 1000 inpatient bed-days, respectively) and a 10.2% increase for ertapenem (5.90 to 6.50 DDD / 1000 inpatient bed-days, respectively). CRE cases were also noted to have significantly been reduced by 33.3% in the medical department post a year after AMS initiation (p < 0.001); with 24 cases reported in 2015 and 16 cases in 2016 [4][9].
Discussion
This study demonstrated that the most common carbapenem used were ertapenem, meropenem and imipenem/cilastatin. Ultimately, approximately of all instances of carbapenem use recorded were considered as non-justified use. Fortunately, however, the AMS-guided intervention performed in the hospital on use of carbapenem prescriptions were generally encouraging, with the most common intervention involving the discontinuation of antibiotic. These findings are congruent to findings from a previous work in a local study conducted at a district hospital [11]. Our results were in contrast, however, to a study conducted in Singapore where the most common intervention involved de-escalation of the therapy towards the use of narrow spectrum antibiotics [12]. In terms of acceptance, the current work demonstrated that de-escalation of antibiotics was the more readily accepted recommendation by the primary team as compared to other recommendations, namely the discontinuation of therapy and changing/escalation of antibiotic. Overall acceptance of recommendations in this study was encouraging and comparable to other similar studies, which also reported an acceptance rate between 70-80% [13][14].
The overall carbapenem consumption was significantly reduced after the introduction of carbapenem stewardship rounds in 2016. This can be attributed to the recommendations under the AMS to discontinue carbapenem or to switch to narrower spectrum antibiotics under appropriate conditionss. These findings were consistent with other similar facilities, where a reduction in broad spectrum antibiotic usage was observed after the initiation of stewardship programs [12][15]. The results of this research depict that in this setting, although there was overall reduction in carbapenem use, an increase in the use of imipenem / cilastatin and ertapenem respectively was observed. The rise in imipenem / cilastatin DDD / 1000 inpatient bed-days in 2016 was mainly attributed to the shift in prescribing pattern by the infectious disease physician in order to either promote antibiotic cycling or balance between meropenem and imipenem / cilastatin use. Meanwhile, the increase in DDD per 1000 inpatient bed-days for ertapenem was also noted to increase in 2016, an occurrence which was attributed to streamlining of meropenem to ertapenem, a carbapenem of a narrower-spectrum, in order to assuage the likelihood of the emergence of MDRO Pseudomonas sp.
In addition to the reduction in carbapenem use, the number of CRE cases was significantly reduced upon initiation of AMS. In support of this finding, a systemic review reported that 11 out of 22 studies on the impact of AMS found significant reductions in antimicrobial resistance upon implementation of AMS [16]. Furthermore, previous work on AMS in a community hospital in United States of America (USA) resulted in a reduction in the use of broad-spectrum antibiotics and a subsequently improved antimicrobial susceptibilities of Pseudomonas sp [17]. However, evidences against these findings exist, another study conducted in a tertiary teaching hospital concluded that although AMS does reduce antibiotics consumption, it does not have any significant improvement on
antimicrobial susceptibilities. Despite that, it is important to note that in the aforementioned study, antimicrobial resistance
was found to have not increased either [18]. These conflicting outcomes may be influenced by the different AMS strategies used, type of healthcare facility and number of intensive care beds involved. Furthermore, it may be that the positive implications of the AMS services implemented on antimicrobial resistance may also take time to be presented more overtly [19], in which case, further work is required to monitor outcomes of AMS in the long term.
Our findings have contributed to the data on the impact of AMS in a tertiary care centre. However, several aspects of our methodology may limit the generalisation of these findings. Firstly, the observational nature of this study renders it vulnerable to influences by confounding factors. In addition, the involvement of a small cohort that includes, only medical wards, would mean the possible existence of type II errors. Thirdly, recommendations on dosage adjustments were not recorded as part of data collection even though it is a routine recommendation during AMS rounds. Finally, carbapenem consumption and CRE cases were analysed through a direct comparison between the data, presented in percentages, of pre-and post-interventions, though it would perhaps be more suitable if an Interrupted Time Series analysis is to be conducted instead with the intention of confirming with greater clarity that the reduction was indeed due to the interventions made. We were unable to carry out this analysis considering the fact that we did not have access to a series of data, in addition to the fact that the intervention only took place during the study period.
Conclusion
AMS-guided interventions of carbapenems appeared to be a useful strategy to reduce non-judicious use of carbapenem in a tertiary hospital and was able to demonstrate a reduction in carbapenem consumption as well as CRE rates in the medical wards. Future long-term studies on the clinical outcomes are required to further investigate the overall impact of the AMS program. Development of a hospital-level AMS policy and promotion of AMS activities via seminars may improve acceptance of AMS interventions in local hospitals.
Acknowledgement
The authors wish to thank the Director-General of Health, Malaysia for the permission to publish this paper. We are grateful to the Hospital Director, Department of Pharmacy and Department of Pathology (Microbiology Unit), Kuala Lumpur General Hospital for their support and contribution towards this research project.
Conflict of Interest
This study has no conflict of interest. This research did not receive any specific grant from funding agencies in public, commercial or not-for-profit sectors.
Reference
- World Health Organisation. Global Report on Surveillance of Antimicrobial Resistance. World Health Organisation. 2014. https://apps.who.int/iris/handle/10665/112642
- Shlaes DM, Gerding DN, John JF, Craig WA, Bornstein DL, Duncan RA, Eckman MR, Farrer WE, Greene WH, Lorian V, Levy S, Mcgowan JE, Paul SM, Ruskin J, Tenover FC, Watanakunakorn C. Society for Healthcare Epidemiology of America and Infectious Diseases Society of America Joint Committee on the Prevention of Antimicrobial Resistance: Guidelines for the Prevention of Antimicrobial Resistance in Hospitals. Clin Infect Dis. 1997 Sep; 25(3):584-99. https://doi.org/10.1086/513766
- Institute for Medical Research. National Antibiotic Resistance Surveillance Report. 2016 https://www.imr.gov.my/images/uploads/NSAR/NSAR_2016/NSAR_report_2016.pdf
- Pathology Department. Hospital Kuala Lumpur Antibiotic Resistance Surveillance Report. 2015.
- Pharmaceutical Services Division Malaysia. Protocol on Antimicrobial Stewardship Program in Healthcare Facilities. 2014. https://www.pharmacy.gov.my/v2/sites/default/files/document-upload/protocol-antimicrobial-stewardship.pdf
- Pharmacy Department. Antibiotic Consumption Surveillance Report. 2015.
- Pharmacy Department. Antibiotic Consumption Surveillance Report. 2016.
- Guidelines for ATC classification and DDD assignment 2018. https://www.whocc.no/atc_ddd_index_and_guidelines/guidelines/
- Pathology Department. Hospital Kuala Lumpur Antibiotic Resistance Surveillance Report. 2016.
- Gaur AH, English BK. The judicious use of antibiotics – An investment towards optimized health care. Indian J Pediatr. 2006;73(4):343–50. https://doi.org/10.1007/BF02825829
- Sing DYF, Boo YL, Mukhlis R, Chin PW, Hoo FK. Antimicrobial stewardship program in a Malaysian district hospital: First year experience. Pakistan J Med Sci. 2016;32(4):999–1004. https://doi.org/10.12669/pjms.324.9855
- Teo J, Kwa ALH, Loh J, Chlebicki MP, Lee W. The effect of a whole-system approach in an antimicrobial stewardship programme at the Singapore General Hospital. Eur J Clin Microbiol Infect Dis. 2012;31(6):947–55. https://doi.org/10.1007/s10096-011-1391-y
- Teng CB, Ng TM, Tan MW, Tan SH, Tay M, Lim SF, et al. Safety and effectiveness of improving carbapenem use via prospective review and feedback in a multidisciplinary antimicrobial stewardship programme. Ann Acad Med Singap. 2015;44(1): 19–25. PMID: 25703493.
- Lew KY, Ng TM, Tan M, Tan SH, Lew EL, Ling LM, Ang B, Lye D, Teng CB. Safety and clinical outcomes of carbapenem de-escalation as part of an antimicrobial stewardship programme in an ESBL-endemic setting. J Antimicrob Chemother. 2014;70(4):1219–25. https://doi.org/10.1093/jac/dku479
- [15] Okumura LM, da Silva MMG, Veroneze I. Effects of a bundled Antimicrobial Stewardship Program on mortality: A cohort study. Brazilian J Infect Dis. 2015;19(3):246–52. https://doi.org/10.1016/j.bjid.2015.02.005
- Nathwani D, Varghese D, Stephens J, Ansari W, Martin S, Charbonneau C. Value of hospital antimicrobial stewardship programs [ASPs]: A systematic review. Antimicrob Resist Infect Control. 2019;8(1):1–13 https://doi.org/10.1186/s13756-019-0471-0
- Day S, Smith D, Harris K, Cox H, Mather A. An Infectious Diseases Physician-Led Antimicrobial Stewardship Program at a Small Community Hospital Associated With Improved Susceptibility Patterns and Cost-Savings After the First Year. Open Forum Infect Dis. 2015;2(2):ofv064. https://doi.org/10.1093/ofid/ofv064
- Cook PP, Catrou PG, Christie JD, Young PD, Polk RE. Reduction in broad-spectrum antimicrobial use associated with no improvement in hospital antibiogram. J Antimicrob Chemother. 2004;53(5):853–9. https://doi.org/10.1093/jac/dkh163
- Jenkins T, Knepper B, Shihadeh K, Haas M, Sabel A, Steele A, Wilson M, Price C, Burman W, Mehler P. Long-term outcomes of an antimicrobial stewardship program implemented in a hospital with low baseline antibiotic use. Infect Control Hosp Epidemiol. 2015;36(6):664–72. https://doi.org/10.1017/ice.2015.41
Please cite this article as:
Anitha Ramadas, Rahela Ambaras Khan, Hwei Lin Teh, Shan Lii Ching, Rohana Hassan, Chee Loon Leong, Khairil Erwan Khalid and Farida Hanim Islahudin, Impact of an Antibiotic Stewardship Program on the Use of Carbapenem in a Malaysian Tertiary Hospital (ACTION). Malaysian Journal of Pharmacy (MJP). 2022;1(8):13-18. https://mjpharm.org/impact-of-an-antibiotic-stewardship-program-on-the-use-of-carbapenem-in-a-malaysian-tertiary-hospital-action/